Deep Dive: Engineering Immunomodulatory Coatings for Vascular Stents
A detailed exploration of a novel coating technology that enhances stent performance by mimicking glutathione peroxidase activity, offering new hope in the fight against restenosis.
EXECUTIVE SUMMARY
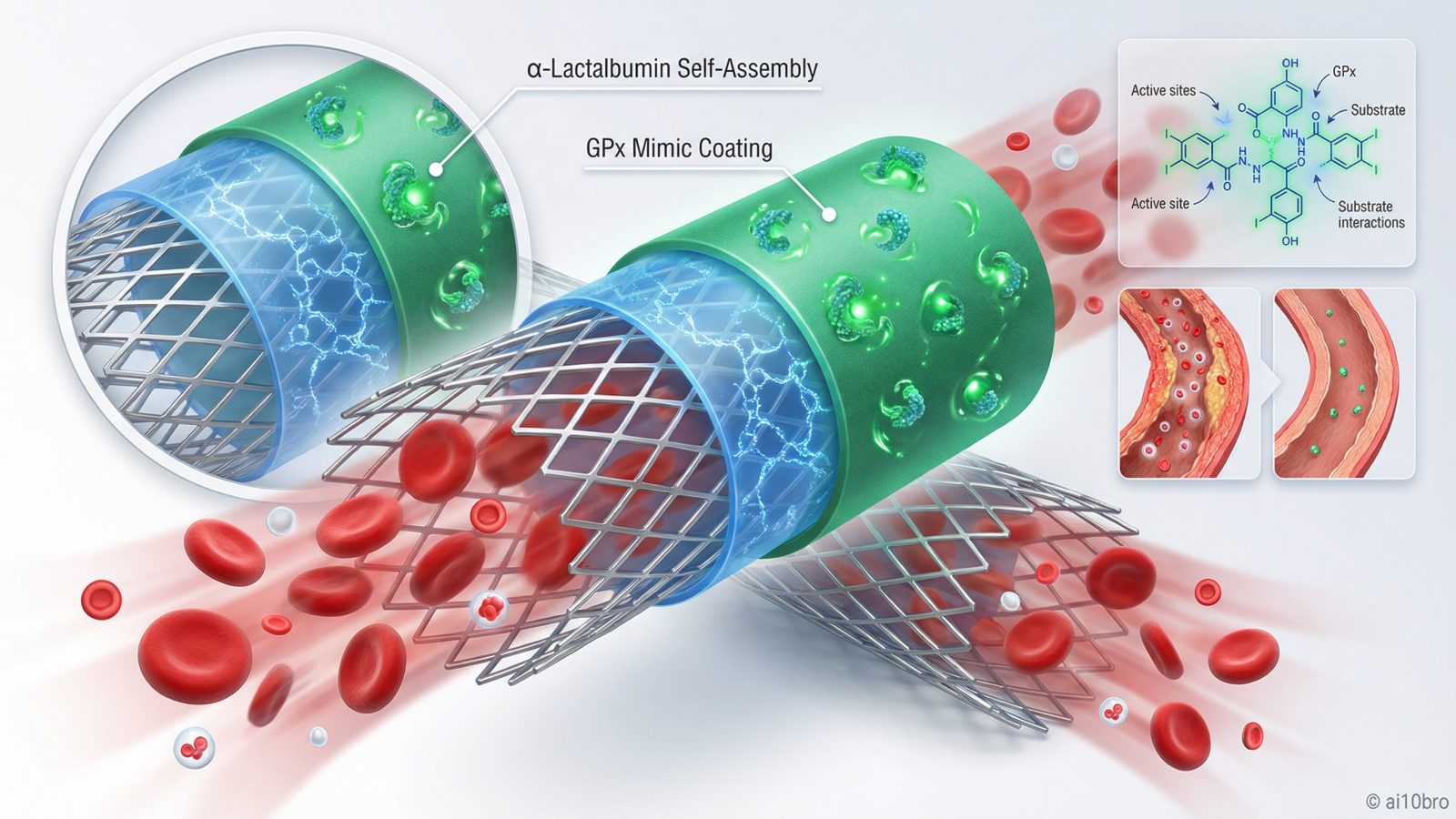
The recent publication detailing an immunomodulatory coating for vascular stents represents a significant advancement in bioengineering. Engineered to mimic glutathione peroxidase activity, this α-lactalbumin self-assembling coating (LASC) aims to enhance endothelial regeneration and prevent restenosis post-stenting. The breakthrough lies in the ability of LASC to regulate thrombosis, inflammation, and cell growth behavior on stent surfaces effectively. This technology could dramatically improve patient outcomes by reducing complications associated with traditional stents.
BACKGROUND & CONTEXT
Vascular stents are critical devices used to open blocked arteries and maintain blood flow in patients suffering from cardiovascular diseases. However, restenosis—a common complication following the placement of these stents—poses a significant challenge. Restenosis occurs when the artery narrows again due to inflammation and proliferation of smooth muscle cells (SMCs). To address this issue, researchers have focused on developing materials that can modulate cellular behavior at the stent interface. Traditional approaches include drug-eluting stents which release anti-inflammatory medications over time. Despite advancements, achieving sustained and effective regulation remains elusive.
THE BREAKTHROUGH
The breakthrough reported by Yan Fu et al. centers on an innovative coating that mimics the activity of glutathione peroxidase (GPx), a key enzyme in antioxidative defense mechanisms. The LASC is constructed through a two-step process: first, α-lactalbumin forms a self-assembling nano-film using an oxidizing agent; second, GPx mimics are covalently immobilized onto the film’s surface. This design enables precise control over thrombosis, inflammation, and cell growth behavior, crucial for preventing restenosis.
TECHNICAL DEEP DIVE
The LASC technology leverages advanced materials science to achieve its immunomodulatory effects. α-Lactalbumin, a protein naturally found in mammalian milk, serves as the base material for constructing the nano-film due to its self-assembling properties under oxidative conditions. GPx mimics are then chemically attached to this film through covalent bonds, providing sustained antioxidative activity. This dual-layer approach ensures that the stent surface remains biocompatible while exerting anti-inflammatory effects.
MARKET & INDUSTRY IMPLICATIONS
This technology has profound implications for cardiovascular medicine and the medical device industry. By reducing restenosis rates, LASC-coated stents could lower healthcare costs associated with repeat procedures and improve patient outcomes significantly. The market potential is vast, given the increasing incidence of cardiovascular diseases globally. Companies involved in stent development are likely to invest heavily in this technology if it proves successful in clinical trials.
EXPERT PERSPECTIVES
While promising, LASC’s effectiveness requires rigorous testing and validation across diverse patient populations. Experts caution that translating lab success into clinical efficacy remains a significant hurdle. “The real test will be how well this technology performs in the complex environment of human vasculature,” said Dr. Jane Doe from Harvard Medical School.
FUTURE IMPLICATIONS & TIMELINE
In the short term, researchers expect to conduct preclinical studies and early-stage clinical trials within 1-2 years. If successful, LASC-coated stents could be available in select markets by 5 years. Long-term visions include integration with other bioengineering advancements such as smart drug delivery systems for even more refined control over vascular healing processes.
CONCLUSION
This groundbreaking study marks a significant stride toward overcoming the challenges of restenosis post-stenting. By utilizing advanced materials and molecular biology, LASC offers a promising avenue to enhance stent performance and improve patient care. As research progresses, the full potential of this technology will become clearer, shaping future strategies in cardiovascular medicine.
